Question
If an orbital electron of the hydrogen atom jumps from the ground state to a higher energy state, its orbital speed reduces to half its initial value. If the radius of the electron orbit in the ground state is r, then the radius of the new orbit would be
Solution
Correct option is
4r
Orbital speed 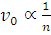 . Therefore, n = 2. Now
. Therefore, n = 2. Now 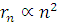 . Hence the radius of the new orbit
. Hence the radius of the new orbit
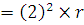
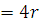 .
.
SIMILAR QUESTIONS
Q1
How many times does the electron go around the first Bohr orbit in one second?
Q2
The electron orbits with principal quantum numbers n > 3 were not allowed, the number of possible elements would be
Q3
According to Bohr’s theory, the energy of an electron in the nth orbit of an atom of atomic number Z is proportional to
Q4
The dimensions of the Rydberg’s constant are
Q5
In terms of the Rydberg’s constant R, the minimum wavelength in the Lyman series is
Q6
The wavelength of the second line of Balmer series is 486.4 nm. What is the wavelength of the first line of Lyman series?
Q7
The wavelength of the first line in Balmer series in the hydrogen spectrumis 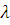 . What is the wavelength of the second line.
. What is the wavelength of the second line.
Q8
The energy needed to ionize the hydrogen atom in the first excited state is 3.4 eV. What is the ionization potential of hydrogen in the ground state?
Q9
The ionization energy of hydrogen atom is 13.6 eV. Hydrogen atoms in the ground state are excited by electromagnetic radiation of energy 12.1 eV. How many spectral lines will be emitter by the hydrogen atom?
Q10
The orbital speed of the electron in the ground state of hydrogen is v. What will be its orbital speed when it is excited to the energy state –3.4eV.

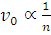
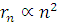
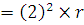
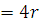 .
.